Sex Differentiation Begins During Early Development
By Claire Asher, on 27 August 2014
Males and females look different from each other, and these sexual dimorphisms are the result, largely, of sex differences in the expression of certain genes. Typically, scientists have studied sexual dimorphism in sexually mature adult animals, as this is the lifestage where differences are most apparent. However, many sex-specific phenotypes arise from sex-biased development, so sex-biased gene expression should be expected to begin during development. Recent research from GEE reveals complex patterns of sex- and stage-dependent gene expression, resulting from differing evolutionary pressures on difference sexes. In fact, sex-biased gene expression is actually most evident during early development.
Although there are some genetic differences between males and females, found on the sex chromosomes (Y in mammals, W in Birds), these contribute relatively little to the physical and behavioural differences between the sexes. Further, some animals (such as many reptiles) don’t possess a sex chromosome at all, instead determining sex based on environmental factors such as temperature. Scientists therefore believe that the majority of sexually dimorphic characteristics are generated from differences in the expression patterns of a shared set of genes, and there is now plenty of evidence for this in a variety of different species. In fact, sexual phenotypes appear to exist along a continuum, with some individuals in certain species exhibiting intermediate traits (e.g. subordinate male Turkeys).

The fruit fly,
Drosophila melanogaster
Juveniles Show Sex-Biased Gene Expression
The authors compared gene expression patterns in pre-gonad tissue in larvae and pre-pupae with gonad tissue in adult flies. Using transcriptome sequencing, which sequences all expressed genes, they were able to detect differences in gene expression between sexes at different developmental stages. By using a single tissue, the authors hoped to gain a clearer picture of gene expression in relation to sex and development, as tissue-specific gene expression can cloud the picture. Gonad and pre-gonad tissue was the obvious candidate, since this tissue forms sperm and eggs and is therefore likely to be under strong selection for sex-biased gene expression.
Despite the long-running assumption that sex-biased gene expression should not be prevalent in juveniles, the results of this study indicated that most genes show sex-bias in at least one pre-adult stage! Over 50% of genes showed at least 2-fold differences in gene expression between the sexes during larval or pre-pupae developmental stages. Sex was still the most important factor, however, with individuals within a sex showing greater similarity in gene expression patterns than individuals within a developmental stage.
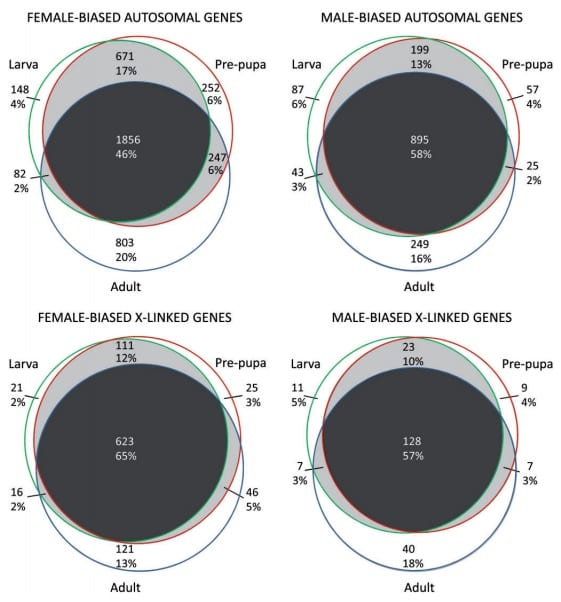
Venn diagrams of the number and percentage of genes showing sex-biased gene expression in larvae, pre-pupae, and adults. Image from open access article.
Continuity and Sex Differences
The majority of sex-biased genes showed expression patterns that remained consistent throughout development, however a significant minority (~25%) of genes showed varying sex-bias according to developmental stage. For example, a gene that showed lower expression in females during the larval stage might then show higher expression in females during adulthood.
In the majority of previous studies have found that more genes show male-bias in adults. By contrast, this study showed that in larval and pre-pupal stages of development in Drosophila melanogaster, more genes show female-biased gene expression. Females were also more likely to show stage-dependent sex-biases in gene expression. The exception to this was genes showing very extreme sex-bias, which tended to be male-biased. This is consistent with the finding that the overall magnitude of gene expression differences tended to be higher in male-biased genes.
The Rate and Form of Evolution
The authors then investigated the evolutionary dynamics that lead to these patterns of gene expression. Genes showing the most rapid recent evolution were those that showed male-biased expression continously throughout life, and those that showed female-biased expression in the larval stages. The evolutionary pressures in male- and female-biased genes were different for each sex. For female-biased genes expressed in larvae, rapid evolution was the result of a relaxation of purifying selection for stage-dependent genes (natural selection that removes harmful mutations), whereas rapid evolution in consistently male-biased genes was a result of stronger purifying selection on stage-dependent genes.
This study reveals complex and intricate relationships between sex, age, development and gene expression in the fruit fly, Drosophila melanogaster. Despite minimal visible differences between the sexes during larval and pre-pupal development, there were vast differences in terms of gene expression. Although this is contrary to previous assumptions about the nature of sex-biased gene expression, it is consistent with the fact that many sexually dimorphic traits exhibited by adults must by necessity begin to develop before adulthood. It is therefore not surprising that sex-biased gene expression is evident in juveniles, however the extent of this bias is quite a surprise. More research is needed to understand the evolutionary dynamics shaping development- and sex-specific gene expression, and how these patterns vary across different tissue types.
Original Article:
Further Reading:
This research was made possible by funding from the European Research Council (ERC), the Elizabeth Hannah Jenkinson Fund and the John Fell Oxford University Press Research Fund
 Close
Close


