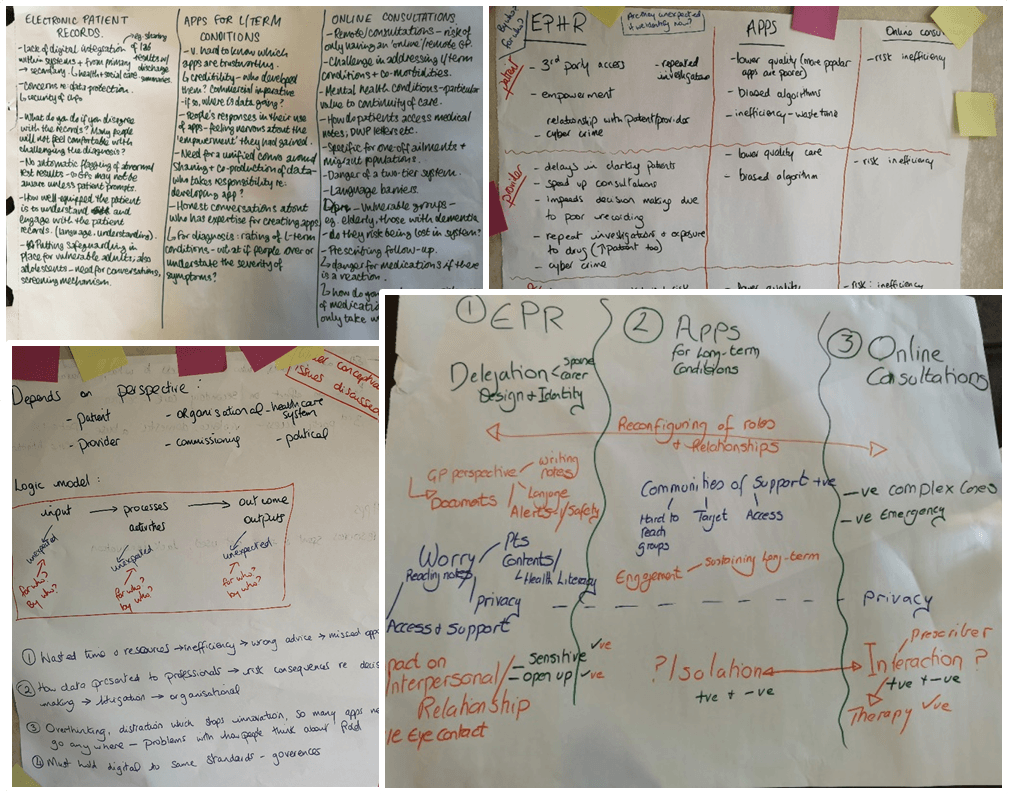
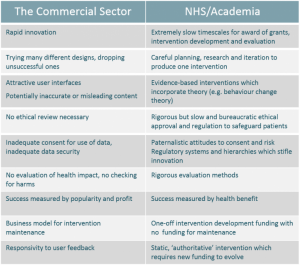
Researcher Tips for Conducting Remote Research in Older Populations
By Megan Armstrong, on 16 December 2021
This blog is written by Tasmin Rookes, Research Assistant for the PD-Care programme. Tasmin also runs a Research Assistant group – if you’d like more information about this group please email Tasmin here: t.rookes@ucl.ac.uk
Since the COVID-19 pandemic research activity, such as identifying, consenting, and assessing participants has been conducted online and remotely. With many clinical populations being older adults, without consistent levels of technology knowledge, this has introduced some challenges. Reflecting on the PD-Care feasibility study, I share our experience and learnings of conducting remote research with people with Parkinson’s.
Identifying potential participants
On the morning of clinics, send reminder emails to the clinical teams to remind them to approach potentially eligible participants with a copy of the participant information sheet.
When following up with a phone call to discuss further, have a loose script with the key information points participants need to know, including the next steps involved to get them enrolled.
Where relevant, recruit or include participants carers or family members to assist with explaining the study, setting up technology or assisting with future research activity, particularly if the person has mild cognitive impairment.
Consenting participants
If obtaining consent online, follow-up with phone calls or emails to reminder people to complete the consent form and keep it at the forefront of their mind. Older people often aren’t used to managing an email inbox, so sometimes resending the link to the consent form can help.
If obtaining written consent, via the post, follow-up after one week of posting to determine if it has arrived and if they have any questions. Be prepared for postal delays and always offer to resend out if participants have waited a long time.
If obtaining verbal consent, ensure you have posted or emailed a copy of the consent form, so participants can follow along. Ensure they are engaged by asking them to state their name and date for the recording. Read through each statement clearly, asking participants to confirm they consent to each statement. Be prepared for questions and consider that your answers are being audio recorded. Save audio recording in a secure location and complete a consent form stating verbal consent was obtained, to send a copy to participants, GPs, and upload to patient notes.
Completing assessments
Be aware of potential risk and safeguarding. If a participant discloses something concerning, follow the guidance in the study protocol and ask for advice from senior colleagues. Keep records of all decision making and collect the participants GP and clinical team contact details for future use.
Completing assessments can be lengthy and is more tiring when completed on a screen. Be transparent about how long they will take and offer comfort breaks or break into shorter sessions where appropriate. Also, providing a written copy of the assessments to participants can help them to follow along with the questions as they are being asked. Allow plenty of time for the assessment and give participants time to think and respond.
Consider whether the assessments you want to do can be completed remotely. If not, consider self-report measures or look for measures that have been adapted to be delivered remotely.
It is inevitable that some assessment appointments will be missed by participants. Send reminder letters, emails, or phone calls to prevent this from happening and provide them with your contact details, so they can inform you if they can no longer attend. Try to reschedule as soon as possible, particularly for follow-up assessments where timelines can be tight.
Be prepared for technical issues and understand how to talk someone through joining calls using the chosen software. Get an understanding of how confident they are, so you can support them and factor this into the time allocated.
Key messages
Most people are happy to conduct research remotely and on the whole older populations are comfortable using technology if they are supported.
Have a clear process in place and test with the team or PPI members before starting to recruit, to iron out any potential issues.
Allow plenty of time at every step, being patient, friendly and calm. You may be the only person they speak to that day, or they may be anxious about using the technology or completing the assessments, so this may help put them at ease.
Consider all potential impairments participants may have, including vision, hearing and speech, and have strategies in place to overcome these and be inclusive. These include, offering study documents in large print, providing contact through email or letter, and having a carer or family member present to support.
Be organised, keep logs of what has been done and what is still to do, so recruitment and follow-up schedules are adhered to.
As we develop more digital interventions and conduct research activity remotely, adapting our approach and using the tips outlined above, successful research conducted remotely with older populations is not only feasible, but can be successful and beneficial.
 Close
Close