The Transcriptional Profile of A ‘Wingman’
By Claire Asher, on 27 November 2013
In many species, males have special adaptations to attract females. From antlers to stalk-eyes, to bright plumage and beards, males across the animal kingdom work hard to look attractive to the opposite sex. In some species, looking good isn’t enough, though. Male wild turkeys need a less attractive ‘wingman’ to help him attract a woman. Wingman turkeys show less extreme sexual traits than the dominant males, and researchers from GEE have exploited this ‘intermediate phenotype’ to investigate how gene expression controls sexual traits. Subordinate male turkeys are physically demasculinised, and this is reflected in their gene expression. Whilst clearly male, these turkeys tend to express ‘male’ genes less and ‘female’ genes more, indicating that sexual phenotypes, at least in this species, exist upon a continuum. This has important implications for how we view sexual selection and sexual traits across the animal kingdom, as well as in our own species.
You might think this is a face only a mother could love, but to a female turkey, this guy is very attractive. Male wild turkeys have a number of physical traits that make them very, very sexy…. to female turkeys, at least. They are larger than females, their feathers are iridescent, their faces are bright red and adorned with caruncles, wattle and a snood. As they grow up, young male turkeys argue with their brothers for dominance. One brother wins, and the others become subordinate. These subordinate males rarely get the chance to mate themselves, but helping out their brother means passing on their genes indirectly. If, later in life, the dominant brother dies, the subordinate may get his chance in the limelight and develop a dominant phenotype. This shows just how plastic sex can be in the animal kingdom.
Differences in Gene Expression
Differences between the sexes form ‘sexual phenotypes’ that are largely the result of the differential expression of genes that are present in both sexes. Although sex chromosomes are also important in controlling the development of sexual traits, many genes on other chromosomes also contribute. Many genes show differences in expression between the sexes.
Theory suggests that genes expressed differently in different sexes are responsible for generating the sex-specific characteristics we observe. However, this hypothesis has been difficult to test in species where sex is binary. This is where wild turkeys come in handy – the existence of the subordinate male phenotype, with reduced sexual traits, provides a rare opportunity to decouple genetic sex and sex-biased gene expression. If sex-biased genes encode sex-specific phenotypes then the subordinate male phenotype should be expected to be the product of reduced expression of male-biased genes and increased expression of female-biased genes.
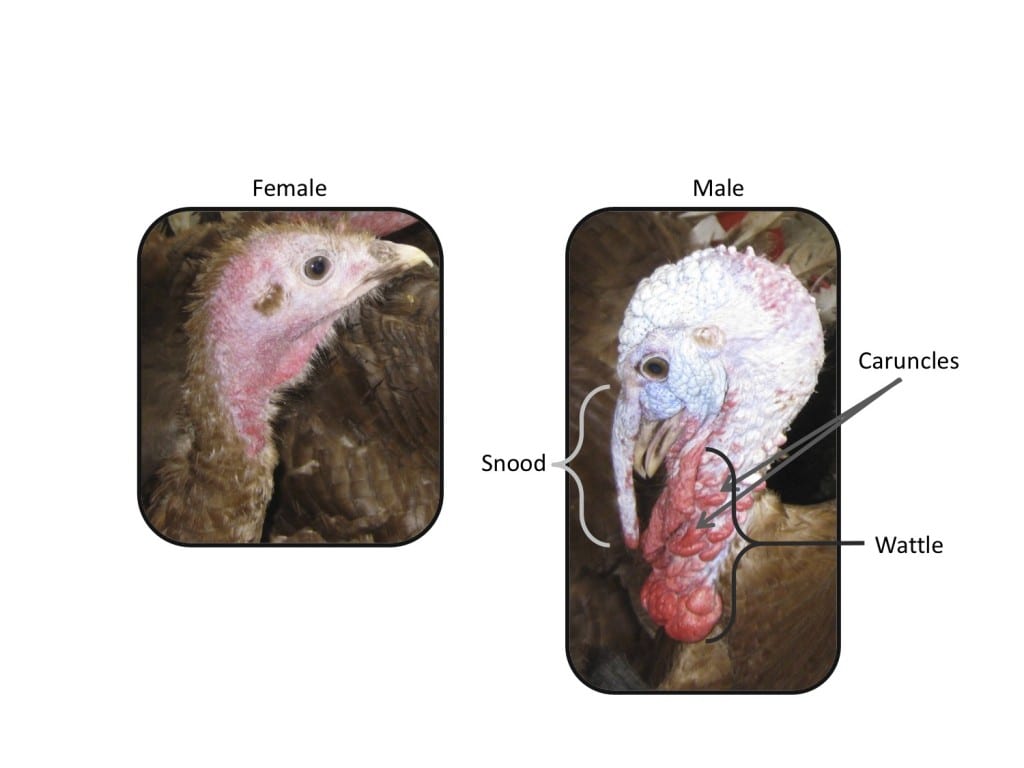
Researchers in GEE and the Department of Zoology, University of Oxford, investigated gene expression patterns in females, dominant and subordinate males. Around 2000 genes were male biased and nearly 3000 showed female bias.
Unravelling the ‘Wingman’ Phenotype
The gene expression patterns of subordinate males clearly clustered with dominant males. So subordinates are not intersex. However, there were subtle differences between the subordinate and dominant males. Subordinate males tended to express male-based genes at lower levels than dominant males, indicating transcriptional demasculinisation. They also expressed female-biased genes at a higher level than dominant males, suggesting transcriptional feminisation. Expressionally, subordinate males were less masculine and more feminine than dominant males. This is consistent with their visual appearance.
Furthermore, genes that were more strongly sex-based showed a greater level of demasculinisation in subordinates than those that were less strongly sex-biased. This suggests that the genes we find to be most strongly sex-biased may contribute the most to generating the male or female phenotype.
These patterns were also true for genes found on the z chromsome – the sex chromosome in birds (analogous to our ‘Y’ chromosome). However, the patterns were no stronger for these genes than for those on other chromosomes, suggesting that the sex chromosome does not contribute disproportionately to the male or female phenotype. This might come as a surprise – surely the sex chromosome should control sex? There is now a great deal of evidence that this isn’t the case, and many animals get on just fine without a sex chromosome at all!
Dominant and subordinate males reveal a continuum of sexual characteristics physically, and this is mirrored by gene expression patterns. Whilst clearly male, subordinate males showed up-regulated of ‘feminine’ genes and down-regulation of ‘masculine’ genes across the transcriptome. This demonstrates for the first time that sex-biased genes are indeed important in generating the sexual traits we observe in nature. Physical sex is determined by the combined action of many genes, some male-biased and some female-biased, and intermediate physical characteristics are possible simply through altering the expression of key sex-biased genes. This result has clear implications for how we view sex and gender in our own species, and greatly enhances our understanding of sexual selection and sexual dimorphism across the animal kingdom.
Original Article:
This research was made possible by funding from the European Research Council
 Close
Close









